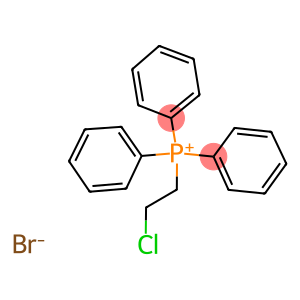
(2-CHLOROETHYL)TRIPHENYLPHOSPHONIUM BROMIDE
2-chloroethyltriphenylphosphonium bromide
CAS: 31238-20-7
Molecular Formula: C20H19BrClP
(2-CHLOROETHYL)TRIPHENYLPHOSPHONIUM BROMIDE - Names and Identifiers
| Name | 2-chloroethyltriphenylphosphonium bromide |
| Synonyms | 2-chloroethyl(triphenyl)phosphanium 2-Chloroethyltriphenylphosphonium bromide 2-chloroethyltriphenylphosphonium bromide (2-CHLOROETHYL)TRIPHENYLPHOSPHONIUM BROMIDE |
| CAS | 31238-20-7 |
| EINECS | 250-527-7 |
| InChI | InChI=1/C20H19ClP.BrH/c21-16-17-22(18-10-4-1-5-11-18,19-12-6-2-7-13-19)20-14-8-3-9-15-20;/h1-15H,16-17H2;1H/q+1;/p-1 |
(2-CHLOROETHYL)TRIPHENYLPHOSPHONIUM BROMIDE - Physico-chemical Properties
| Molecular Formula | C20H19BrClP |
| Molar Mass | 405.7 |
| Melting Point | 169-172°C |
| Sensitive | Hygroscopic |
(2-CHLOROETHYL)TRIPHENYLPHOSPHONIUM BROMIDE - Risk and Safety
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. |
(2-CHLOROETHYL)TRIPHENYLPHOSPHONIUM BROMIDE - Introduction
The bromide is an organic phosphorus compound with the chemical formula C16H15ClPBr. The following is a description of the properties, uses, preparation and safety information of the compound:
Nature:
bromide is a white crystalline solid with a benzene-like aroma. It has a melting point of about 150-154 degrees Celsius and is soluble in common organic solvents such as ethanol, chloroform and dimethyl sulfoxide.
Use:
is widely used as a reagent in organic synthesis. It can be used as a methylating agent and is often used in methylation reactions, for example, in the synthesis of organic phosphorus compounds. It can also be used for nucleophilic substitution reactions, oxidation reactions and addition reactions, etc.
Method:
The preparation method of bromide can be achieved through the following steps: First, triphenylphosphine and or diphenylthionyl chloride are reacted under appropriate reaction conditions to synthesize (phosphonium) triphenylphosphonium bromide, it is then reacted with vinyl chloride in a suitable solvent to give the desired product.
Safety Information:
The bromide is a compound that may be harmful to the human body, so you need to pay attention to safety measures during operation. Direct contact with skin and eyes should be avoided, and the operation should be carried out in a well-ventilated place. During handling and storage, contact with oxidants, strong acids and strong bases should be avoided to avoid dangerous reactions. In the event of an accident, appropriate first aid measures should be taken immediately and medical professionals should be consulted.
Nature:
bromide is a white crystalline solid with a benzene-like aroma. It has a melting point of about 150-154 degrees Celsius and is soluble in common organic solvents such as ethanol, chloroform and dimethyl sulfoxide.
Use:
is widely used as a reagent in organic synthesis. It can be used as a methylating agent and is often used in methylation reactions, for example, in the synthesis of organic phosphorus compounds. It can also be used for nucleophilic substitution reactions, oxidation reactions and addition reactions, etc.
Method:
The preparation method of bromide can be achieved through the following steps: First, triphenylphosphine and or diphenylthionyl chloride are reacted under appropriate reaction conditions to synthesize (phosphonium) triphenylphosphonium bromide, it is then reacted with vinyl chloride in a suitable solvent to give the desired product.
Safety Information:
The bromide is a compound that may be harmful to the human body, so you need to pay attention to safety measures during operation. Direct contact with skin and eyes should be avoided, and the operation should be carried out in a well-ventilated place. During handling and storage, contact with oxidants, strong acids and strong bases should be avoided to avoid dangerous reactions. In the event of an accident, appropriate first aid measures should be taken immediately and medical professionals should be consulted.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: (2-Chloroethyl)triphenylphosphonium bromide Request for quotation
CAS: 31238-20-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 31238-20-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: (2-Chloroethyl)triphenylphosphonium bromide Request for quotation
CAS: 31238-20-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 31238-20-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
View History
